 
Inhalers can have generic names and be produced by different drug companies too. There are many different brands of inhalers. 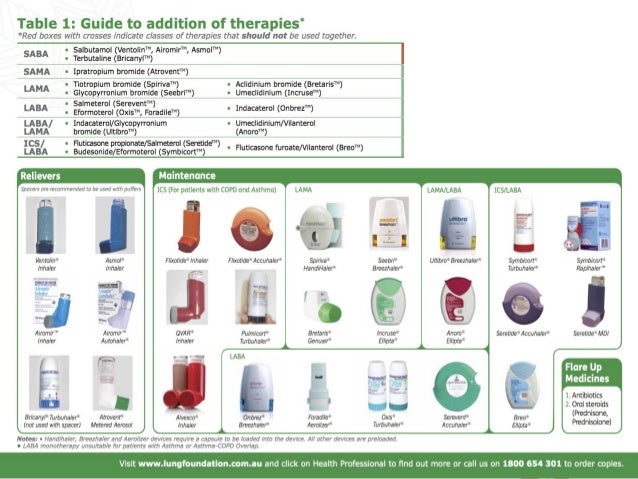
Different drug companies can use the generic medicine and produce different brands - the proprietary medicine names. The proper medicine name is called the generic name. The airways and lungs are treated but little of the medicine gets into the rest of the body (via the bloodstream). This means that you need a much smaller dose than if you took the medicine as a tablet or liquid by mouth. The medicine inside an inhaler goes straight into the airways when you breathe in. There are also separate leaflets called Asthma and Inhalers for Asthma. For general information about COPD, see the separate leaflet called Chronic Obstructive Pulmonary Disease. The only common side-effect was dry mouth, which can be attributed to the anticholinergic properties of tiotropium.This leaflet is about inhalers for chronic obstructive pulmonary disease (COPD). The safety and tolerability of the combination were similar to that of tiotropium or olodaterol alone. In addition, reductions in the use of rescue medication were observed in patients treated with the combination compared with those who used tiotropium alone (22% and 26% improvement in use of daytime and night-time rescue salbutamol, respectively) or olodaterol alone (13% and 18% improvement in use of daytime and night-time rescue salbutamol, respectively) (p<0.0001 for all comparisons). Patients who used the combination showed significant reductions in breathlessness (as measured by a clinically meaningful improvement in focal Transition Dyspnoea Index score) compared with those who used tiotropium or olodaterol alone (54.9% vs 50.6% or 48.2% ). More patients experienced noticeable improvements in their quality of life after 24 weeks (as indicated by a reduction in St George's Respiratory Questionnaire total score) with the combination than with tiotropium or olodaterol alone (57.5% vs 48.7% or 44.8%, respectively). In both studies, improvement in lung function (as assessed by FEV 1 AUC 0-3h and trough FEV 1 responses) after 24 weeks was significantly greater in patients treated with tiotropium/olodaterol (5 microgram/5 microgram, n=522) than in those treated with tiotropium alone ( Spiriva) (5 microgram, n=526) or olodaterol alone ( Striverdi Respimat) (5 microgram, n=525) (p≤0.0001 for all comparisons). All study medications were administered once daily in the morning via the Respimat inhaler. The efficacy and safety of the tiotropium/olodaterol combination were assessed in two 52-week, double-blind, parallel-group phase III studies comparing use of the combination with its individual components in patients with moderate to very severe COPD. 
The combination of a LAMA and a LABA thus provides additive bronchodilatation in all regions of the lungs. Muscarinic receptors are more prominent in the central airways and β2 adrenoceptors have a higher expression level in the peripheral airways. Each puff delivers 2.5 microgram tiotropium and 2.5 microgram olodaterol the recommended dose is two puffs once daily. Spiolto Respimat is indicated as a once-daily maintenance treatment to relieve symptoms in adults with moderate to severe chronic obstructive pulmonary disease (COPD). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
